INTERACTIVE PERIODIC TABLE OF ELEMENTS
BRINGING THE PERIODIC TABLE TO LIFE
INL INTERACTIVE PERIODIC TABLE OFFERS DETAILED INFORMATION ON ALL 118 ELEMENTS
Idaho National Laboratory has enlisted its experts, researchers and writers to produce an online interactive Periodic Table that offers pop-up information on every single known element. The feature is a testament to the scientific research done at the lab, but also ties into INL’s education and outreach mission, which stretches across Idaho to the most remote and rural places. This mission is evolving all the time, providing relevant and engaging content for teachers and resources for students seeking help with STEM subjects.
Click below to discover information about each element.
*Information for this chart was provided by INL experts along with multiple online sources.
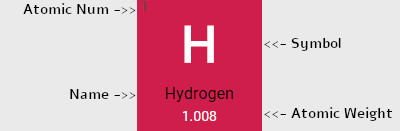
- Subcategory in the metal-metalloid-nonmetal trend
- Alkali Metal
- Alkaline Earth
- Nonmetal
- Metalloid
- Post-Transition Metal
- Transition Metal
- Halogen
- Noble Gas
- Lanthanoids
- Actinoids
Favorite Elements at INL
Check out the quotes below to learn what draws some current and former employees to certain elements.
















HISTORY OF THE PERIODIC TABLE
There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.

Attempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight.
Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists.
The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. (In the modern periodic table, a group or family corresponds to one vertical column.)
The Periodic Table
The Periodic Table: Crash Course In Chemistry
By Crash Course 11:21min
Hank gives us a tour of the most important table ever, including the life story of the obsessive man who championed it, Dmitri Mendeleev. The periodic table of elements is a concise, information-dense catalog of all of the different sorts of atoms in the universe, and it has a wealth of information to tell us if we can learn to read it.
The genius of Mendeleev’s periodic table
By TEDed 4:24 min
The elements had been listed and carefully arranged before Dmitri Mendeleev. They had even been organized by similar properties before. So why is Mendeelev’s periodic table the one that has endured? Lou Serico explains via eka-aluminum, an element whose existence Mendeelev predicted years before it was discovered.
Daniel Radcliffe raps “The Elements”
By The Graham Norton Show 1:38min
Harry Potter star Daniel Radcliffe performs his party trick, which is singing “The Elements” by musical humorist Tom Lehrer.
The Race to Invent the Periodic Table
By PBS Digital Studios 5:43min
Some chemists might see the periodic table of elements as a holy testament to the power of science. However, when it first debuted, it was a different kind of holey, and its journey to classroom walls everywhere had a whole lot of bumps. Watch as Reactions digs into the history of the periodic table with the help of a vanishing spoon, a man named after a rooster, and a bearded Russian.