Hydrogen is all around us but out of sight, tied up in water, natural gas, plastics and elsewhere. Freed, that hydrogen could power cars and store energy for use later – all while leaving no emissions behind but water.
What’s more, hydrogen, the lightest and most abundant element in the universe, is already important to a range of vital and strategic industries.
“You use hydrogen in the production of steel. You use hydrogen in the support of new types of vehicles, electric vehicles. You use it for the production of fertilizers,” said Bruce Hallbert, director of the U.S. Department of Energy’s Light Water Reactor Sustainability Program, based at Idaho National Laboratory (INL). “So, there’s a very large demand.”
However, current commercial hydrogen production has a significant carbon footprint. Now, projects funded by DOE aim to change that by exploiting the capabilities of nuclear power plants. Three utilities – Akron, Ohio-based FirstEnergy Solutions, Xcel Energy of Minneapolis, and Arizona Public Service (APS) of Phoenix – will be rolling out projects over the next few years that demonstrate technology to make hydrogen from water on an industrial scale. Hallbert’s group at INL is also participating, having worked on the concept for years. If successful, the result of the projects will be more hydrogen with no carbon emissions for the world at large and a new revenue stream for the utilities.
This scenario sets the stage for the pilot projects. A collaboration between DOE, national labs, EPRI and industry, the projects are the result of a multiyear planning process. They’re designed to demonstrate the viability hydrogen production via nuclear powered electrolysis.
Significant as it is now, the opportunity presented by hydrogen could be much larger in the future.
“Hydrogen has the highest energy content by weight of all known fuels,” Hallbert said. That means it can be used to store and transport energy, either to move cars or power houses and buildings.
Right now, there are thousands of hydrogen fuel cells in commercial vehicles, forklifts, and backup power units throughout the United States. To get an idea what the upper limit might be if hydrogen takes off as a fuel, consider that car and truck sales in the U.S. run about 16 to 17 million vehicles annually. If a significant fraction of those were hydrogen powered, then the total demand for the fuel would be substantially higher than it is now.
But for that future shift to happen, many parts of infrastructure must be put into place, starting with the fuel itself.
Fixing current production drawbacks
Today, approximately 60 to 70 million metric tons of hydrogen are produced worldwide, with a sixth of that made in the U.S. Hydrogen is industrially manufactured using a process known as steam reforming. This begins with a fossil fuel, such as natural gas. Steam reforming splits the hydrocarbon into free hydrogen and carbon. The former is collected while the latter combines with oxygen in the air to form carbon dioxide. Since natural gas is mostly methane, or CH4, there is roughly one carbon dioxide, CO2, molecule emitted for every two molecules of hydrogen, H2, generated.
But there are ways to make hydrogen without carbon emissions, such as through electrolysis. Passing a current through water separates it into hydrogen and oxygen. This can be done more efficiently by raising the temperature using steam. The heat of the steam then supplies some of the energy needed to free the hydrogen, reducing the amount of electricity required.
“High temperature electrolysis is more efficient, which means you get more hydrogen from it for a given amount of energy,” Hallbert said.
This process typically is done with steam at a temperature ranging from 100o C (212o F) to as high as 850o C (about 1,550o F). Selecting the temperature of the steam involves, among other things, balancing the efficiency gained against the increased cost.
The electricity that powers the electrolysis can come from any source, including carbon-free solar, wind or nuclear power plants. Electrolysis benefits from lower electricity costs, and the price of producing power carbon free continues to fall. The International Renewable Energy Agency predicts that renewables will be a less expensive source of electricity than any fossil fuel by 2020.
That good news is tempered by the reality that the sun doesn’t always shine, and the wind doesn’t always blow. This means there must be some means to supply electricity when that happens, one role that nuclear plants play today.
At other times, the sun is out in full force and the winds strong. The effect of these changing conditions is that the grid can sometimes have too much electricity. Since power can’t currently be economically stored, the solution is to idle some production, which may include any nuclear plants.
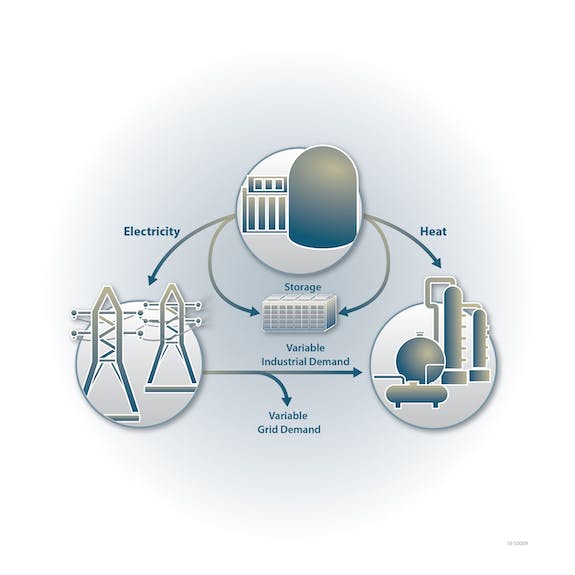
Hydrogen production offers a way to soak up extra generating capacity when it is present. That can improve the economics of the power production. In this, nuclear plants have an advantage because they produce both electricity and steam. Thus, low- or high-temperature electrolysis facilities could be located adjacent to or near a nuclear plant. This would allow hydrogen to be made during those times when capacity is available.
“When we have surplus energy on the electrical grid, instead of ramping down a nuclear unit, we could shift that energy to hydrogen production. That would result in a higher capacity factor and would maximize the utilization of carbon-free power sources,” said Tim O’Connor, chief nuclear officer for Xcel Energy.
Supplying proof
So, hydrogen production looks promising. There’s current demand and the possibility of a much larger market in the future. Also, renewables are dropping the cost of electricity, which makes electrolysis increasingly competitive as compared to alternative hydrogen making methods. Finally, renewables are adding a growing element of production variability, which means that there will be excess capacity that could be used to make hydrogen.
Paving the way for this scenario begins with a two-year project from FirstEnergy Solutions, which will use a low-temperature electrolysis unit to produce commercial quantities of hydrogen for public transportation fleets, steelmaking and other applications. In a release, Raymond Lieb, senior vice president of fleet engineering, signaled eagerness to begin.
“This is a great opportunity to show that hydrogen can be effectively generated in a carbon-free and safe manner,” he said of the project, which runs through 2021.
A second demonstration will run at Xcel Energy, which is pursuing 100 percent carbon-free electricity production by 2050. O’Connor said the hydrogen project will help ensure the company’s nuclear power plants continue to support Xcel Energy in providing reliable, carbon-free electricity.
The third pilot will be at an APS nuclear plant near Phoenix and will employ advanced hydrogen production technologies, such as high-temperature electrolysis.
“This project allows us to explore a new form of energy storage while continuing to provide customers what they want – clean, affordable and reliable electricity,” said Bob Bement, APS executive vice president and chief nuclear officer, in a release announcing the project.
INL’s Hallbert noted that success in these demonstrations will confirm that commercial nuclear utilities can produce essential products like hydrogen without carbon emissions. Doing so, he said, could enable steel producers to reduce their carbon footprint by as much as 15 percent and help fertilizer manufacturers substantially cut carbon emissions.
“As you start to look into the future, it’s very feasible to scale up these types of technologies and produce significant quantities of hydrogen,” Hallbert said of the methods found in the pilot projects. “Commercial nuclear power plants could start to address some large portion of that demand for hydrogen.”
So, these demonstrations can provide a new revenue stream for utilities. They also can help pave the path to a hydrogen economy, one where fuel is made from and turns back into water in a cycle free of carbon emissions.