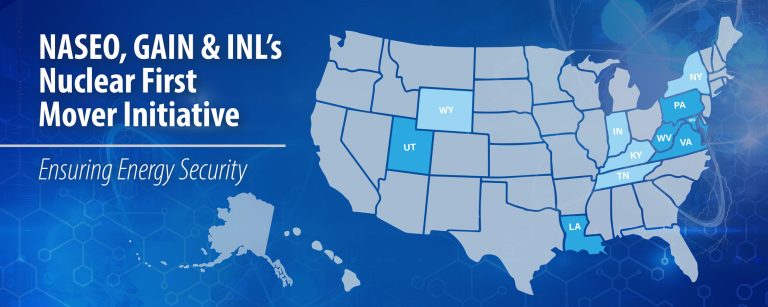
As materials heat up and cool down, atoms and molecules can rearrange in strange and interesting ways.
Water is one example. While most materials get denser when they freeze, water forms a crystalline structure as a solid that is less dense than its liquid form. That’s why ice cubes float.
And these changes aren’t limited to the atomic and molecular level — structural changes, visible to the naked eye, can also occur.
Understanding how atoms and molecules behave during heating and cooling is especially important for developing the metals, ceramics and alloys used to make nuclear fuels and materials.
For the first time in decades, researchers at Idaho National Laboratory are studying how the structure of uranium changes when it is rapidly heated or cooled.
Surprising behaviors
Experts at INL’s Irradiated Materials Characterization Laboratory — including Fidelma Di Lemma, Andrea Jokisaari, Luca Capriotti, Tiankai Yao and Xiang Liu — used uranium samples that had been bombarded with radiation during an earlier experiment at INL’s Transient Reactor Test Facility for their analysis. While each irradiation had the same total energy release, differences in irradiation time resulted in the samples undergoing different temperature ramps and burnups.
Researchers then used a focused ion beam to slice the samples ultra-thin so that electrons from a transmission electron microscope could penetrate them and get an image.
The researchers equipped the microscope with a special sample holder that heated the uranium to temperatures ranging from room temperature to 850 C.
The results were surprising, including changes that occurred at much lower temperatures than expected. Data from the experiment will help improve the performance and safety of nuclear fuels and materials.
“The last time this was studied was in the 1960s, and the techniques we have now weren’t available,” said Jokisaari, a computational scientist at INL. “We had a lot of questions we wanted to answer.”
An unprecedented look during heating and cooling
When examined under a transmission electron microscope in real time, the samples gave INL researchers an unprecedented look at the changes in structure during heating and cooling.
“Uranium has a really weird thermal expansion behavior,” Jokisaari said. “It expands in two directions and it shrinks in another direction.”
The researchers reported some surprising results that may help explain why uranium acts the way it does. “This material is frisky,” Jokisaari said. “The uranium started changing at much lower temperatures than expected, depending on how fast the material was heated.”
The amount of heat applied had a significant impact on the way each sample behaved, she said.
The three irradiation durations also affected the uranium in different ways.
“The biggest difference was the degree of microstructural damage,” Di Lemma said. “The way each sample responded to heating varied considerably. Some of the material behaved as if it hadn’t been irradiated, and we saw some porosity develop in the samples that we were not expecting.”
Defects in the crystalline structure
The researchers specifically analyzed how defects in crystalline structures formed in the uranium.
One type of defect occurs where crystals intersect with one another. Called the “grain boundaries,” these intersections move as a fuel is heated, creating defects. Other types of defects occur when atoms rearrange within the crystal structure itself. Both types of defects can affect a fuel’s performance.
“The defects in fuels and materials don’t come from just the irradiation, but also how the sample was fabricated,” Di Lemma said.
Even though uranium metal is rarely used in its pure form, understanding how heating a sample affects its structure at different scales — from atoms and molecules to changes visible to the human eye — guides understanding of a material’s fundamental properties.
For example, when conventional nuclear fuel rods heat up, the fuel typically expands. During manufacturing, heating or cooling a nuclear fuel or material too rapidly can lead to a structural weakness.
Predicting how fuels behave in a reactor
With a more complete understanding of uranium dynamics, researchers can develop models and simulations that can better predict how fuels will behave in a reactor. Changes in dimension, porosity, thermal properties and the geometric stability of the fuel can have a big impact on performance. Physics-based modelling enables engineers to design new types of fuel more efficiently.
“If we can use the fuel for longer periods and more effectively manage fuel swelling issues, we can cut down on cost and waste,” Jokisaari said.
In the future, similar experiments on different materials could provide more data for the modelling effort. “We want to continue building our knowledge, including by testing a range of materials beyond just uranium,” said Luca Capriotti, a materials and fuels researcher at INL and another member of the team. “These experiments are absolutely vital in helping us answer questions about fuel performance and behavior as we help develop the advanced nuclear fuels of the future.”